- Blog
- Adobe flash professional cs5 release date
- Hypack syringe products
- Safar ki dua arabic hd images
- Open loop plant simulation
- Download danganronpa killing harmony 100 save
- Mazda cx 5 chip tuning
- Camtasia cracken
- Softube tape crack reddit
- Brunswick bristol ii parts
- How to show unit cell frame in crystal maker
- Heliocentric astrology pdf
- Martian movie run time
- Argus monitor femail

The observation dynamics, relating the anesthetic marker and the dynamic state in the effect compartment, is assumed to be described by a sigmoid-Emax pharmacodynamic (PD) model ( Meibohm and Derendorf (1997)). Here the state dynamics, driven by the drug infusion, is described by a four-compartment linear pharmacokinetic (PK) model. In this numerical study of CLAD, we consider a specific nonlinear state-space model as the plant model. The success of both PID and LQG, as general control paradigms for CLAD, motivated us to compare them through a numerical experiment in this work.
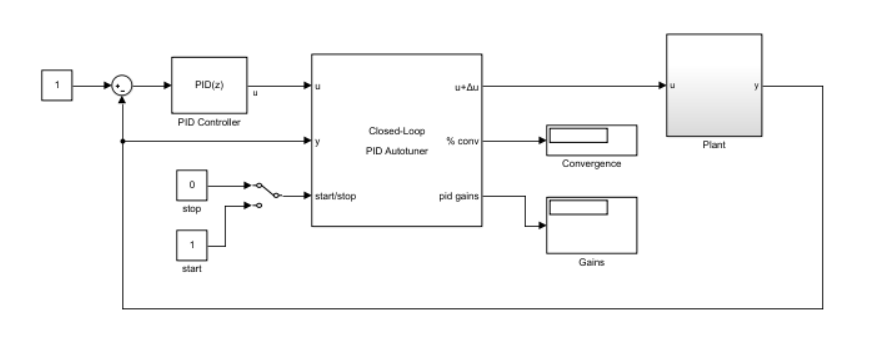
The more recent development of using an estimated-state feedback-based optimal control strategy via linear quadratic Gaussian (LQG) regulation have yielded promising results for maintenance of medically-induced coma in animal models ( Shanechi et al. For example, the classical output-feedback proportional-integral-derivative (PID) control strategy and its variants have been found to be useful in CLAD applications in human studies ( Dumont et al. The CLAD systems reported in prior research, vary with respect to different features, e.g., regime of anesthesia, anesthetic drug, signal modality, marker definition, control strategy, and plant model, just to name a few. The utility of such autopilot systems are in personalized medicine as they can aid anesthesiologists achieve patient-specific precise drug-dosing efficiently, even under resource constrained environments ( Dumont and Ansermino (2013) Absalom et al. As this iterative procedure continues over time, the anesthetic marker ideally would approach the user-prescribed set-point. (2007)).Ī typical CLAD system works in a cyclic mode at a fast rate (~ 0.1 s −1) where in each cycle a computer calculates a scalar anesthetic marker from real-time physiological signals (say, EEG or BP) and uses a feedback control strategy to determine the adjustment of drug-dosage for the next cycle. Analogous systems are also available for blood pressure (BP)-based CLAD ( Ngan Kee et al. Such structured drug-dependent changes in the EEG signals have allowed developments of automated EEG-based closed-loop anesthesia delivery (CLAD) systems that can provide precise control of brain states under anesthesia ( Bickford (1950) Schwilden et al. For example, changes in oscillatory patterns in the scalp electroencephalogram (EEG) has been shown to be correlated with behaviorally-defined changes in unconsciousness levels during GA ( Purdon et al. (2010)), conventionally, an anesthesiologist would monitor relevant physiological signals that are known to be associated with anesthetic depth, and manually adjust the drug dose.
During major surgeries performed under general anesthesia (GA) ( Brown et al. More than 230 million patients undergo major surgeries world-wide under anesthesia ( Weiser et al. This paper may also serve as a tutorial paper for teaching control theory for CLAD. The numerical analysis framework and findings reported here can help CLAD developers in their choice of control strategies. In the case of noisy observations, ILQG can be tuned to provide a smoother infusion rate while achieving comparable steady state response with respect to PID. Based on the numerical analyses performed here, conditioned on our choice of model and controllers, we infer that in terms of accuracy and bias PID control performs better than ILQG which in turn performs better than LQG. Specifically, we numerically simulate multiple CLAD sessions for the scenario where the plant model parameters are unavailable for a patient and the controller is designed based on a nominal model and controller gains are held constant throughout a session. For a nonlinear plant model based on well-established models of compartmental pharmacokinetics and sigmoid-Emax pharmacodynamics, we numerically analyze the set-point tracking performance of three output-feedback linear control strategies: proportional-integral-derivative (PID) control, linear quadratic Gaussian (LQG) control, and an LQG with integral action (ILQG). Since control strategies for CLAD vary across the systems reported in recent literature, a comparative analysis of common control strategies can be useful. A typical CLAD system would use an anesthetic marker, calculated from physiological signals, as real-time feedback to adjust anesthetic dosage towards achieving a desired set-point of the marker. Closed loop anesthesia delivery (CLAD) systems can help anesthesiologists efficiently achieve and maintain desired anesthetic depth over an extended period of time.
- Blog
- Adobe flash professional cs5 release date
- Hypack syringe products
- Safar ki dua arabic hd images
- Open loop plant simulation
- Download danganronpa killing harmony 100 save
- Mazda cx 5 chip tuning
- Camtasia cracken
- Softube tape crack reddit
- Brunswick bristol ii parts
- How to show unit cell frame in crystal maker
- Heliocentric astrology pdf
- Martian movie run time
- Argus monitor femail
